Gluten Dairy Enzymes: Using Dairy Enzyme Complex in Industrial Dairy Formulations
Use Dairy Enzyme Complex in cheese, yogurt, and milk processing with pH, temperature, dosage, QC, COA/TDS/SDS, and supplier guidance.

A practical formulation guide for dairy processors evaluating enzyme systems for lactose hydrolysis, milk modification, cheese yield, texture control, and clean-label process optimization.
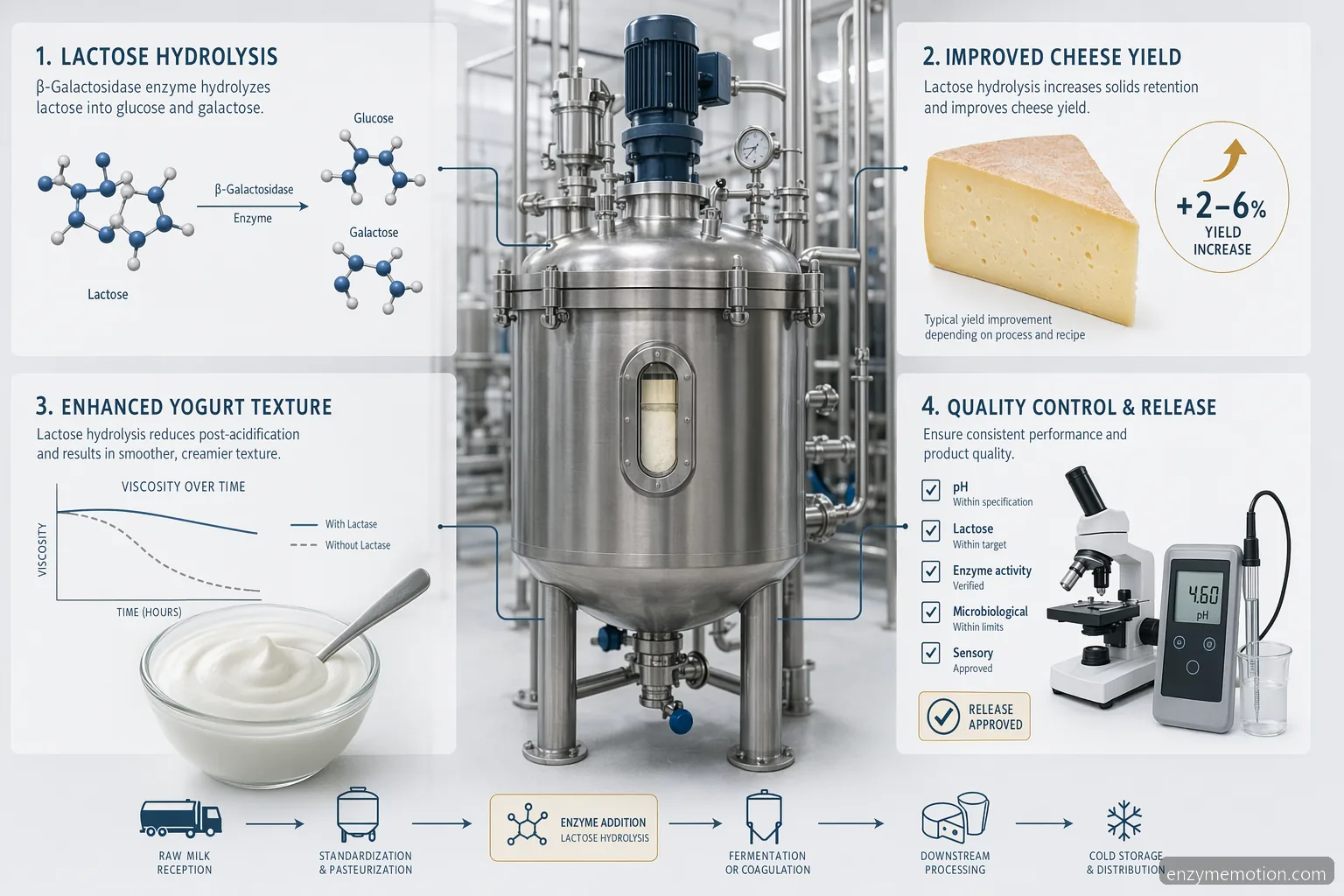
What “Gluten Dairy Enzymes” Means for Industrial Buyers
In B2B dairy processing, the phrase “gluten dairy enzymes” is often used by procurement or R&D teams looking for enzyme inputs that fit dairy formulations while supporting gluten-control requirements in the production environment. It should not be confused with consumer phrases such as digestive enzymes for dairy intolerance, best digestive enzymes for gluten and dairy intolerance, digestive enzymes for gluten and dairy, gluten free dairy free digestive enzymes, gluten dairy digestive enzymes, gluten and dairy enzymes, or gluten and dairy digestive enzymes. For industrial use, the key question is whether the enzyme preparation is suitable for milk-based substrates, documented for allergen status, and compatible with the target process. Dairy Enzyme Complex may include lactase, protease, lipase, transglutaminase, or microbial coagulant activities depending on the formulation objective. The correct specification is selected by activity profile, carrier system, pH tolerance, thermal behavior, sensory impact, and regulatory fit in the country of sale.
Industrial enzyme use, not supplement positioning • Relevant to cheese, yogurt, and milk processing • Requires documented allergen and gluten-control information • Selected by activity, process fit, and QC endpoint
Application Fit in Cheese, Yogurt, and Milk Processing
For cheese production, dairy enzymes may support coagulation, flavor development, protein modification, or yield management. Microbial coagulants can be evaluated as a rennet alternative where the product brief allows, but performance must be tested against curd firmness, whey clarity, moisture, bitterness, and slice or shred behavior. In yogurt, enzyme systems are often assessed for lactose reduction, texture adjustment, or controlled protein modification before fermentation. In fluid milk processing, lactase is commonly evaluated for lactose hydrolysis under refrigerated or warm incubation conditions, followed by pasteurization or UHT steps where applicable. Formulators should avoid assuming that one dairy enzyme complex can solve every application. A cheese enzyme program may need fast action at milk pH, while a yogurt enzyme system must remain compatible with culture performance, viscosity targets, and post-acidification limits.
Cheese: coagulation, flavor, texture, and yield • Yogurt: lactose reduction and mouthfeel support • Milk processing: lactose hydrolysis and process efficiency • Rennet alternative options require pilot trials
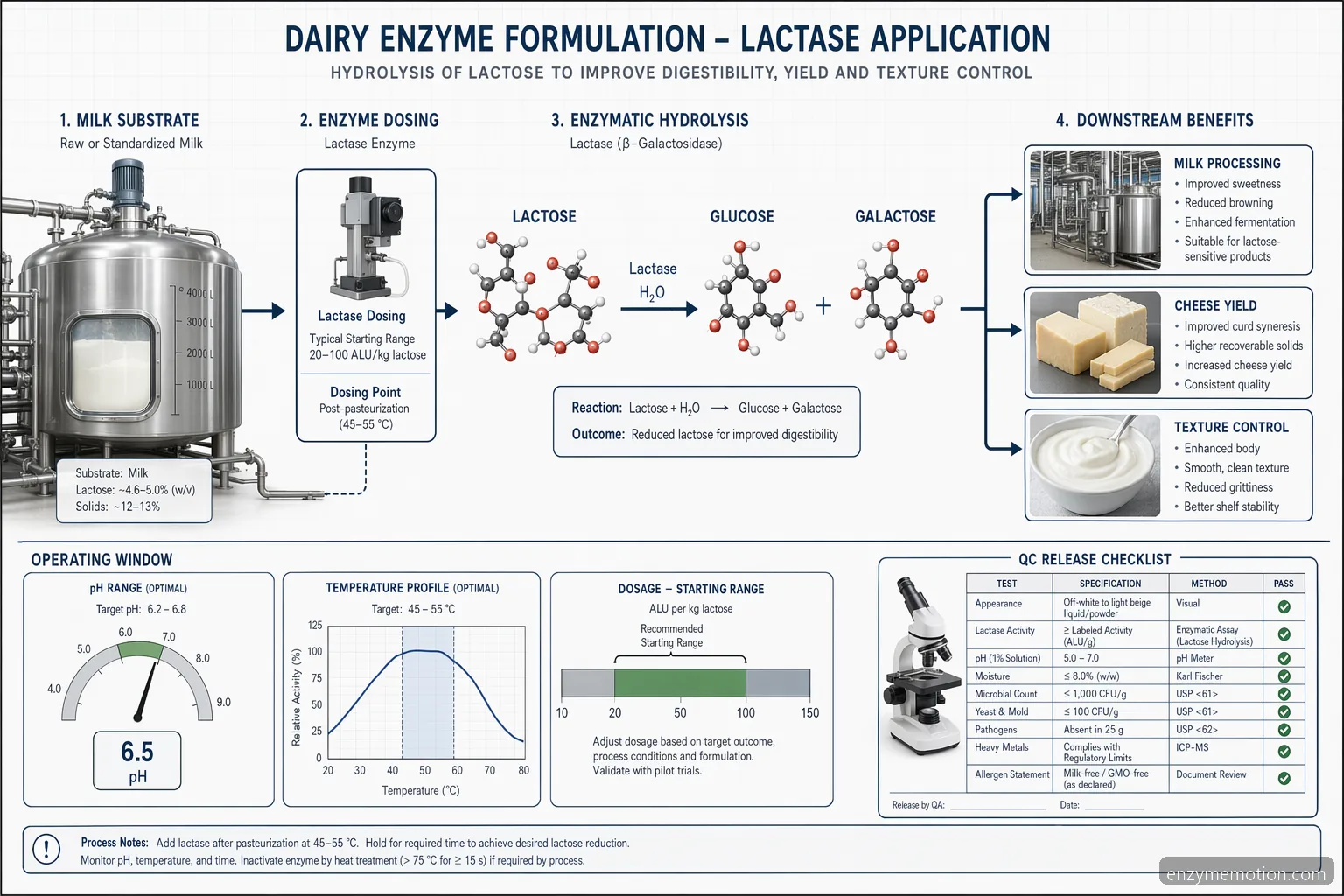
Process Conditions and Dosage Starting Points
Safe starting conditions depend on enzyme activity and product design, so the supplier TDS should always override generic guidance. As a practical pilot range, lactase may be screened around pH 6.4–6.8 in milk at 4–10°C for extended cold hydrolysis or 35–42°C for faster conversion, with dosage commonly explored from 0.02–0.20% w/w depending on activity units and target residual lactose. Coagulant enzymes for cheese are commonly evaluated near milk pH 6.4–6.7 and 30–35°C, with dosage adjusted to achieve target flocculation and cut time. Proteases or lipases require tighter sensory control because overuse can create bitterness, rancidity, or texture defects. Pilot design should include a no-enzyme control, two to three dosage levels, defined hold times, and an inactivation or downstream kill step where required by the process.
Confirm pH and temperature on the product-specific TDS • Use activity units, not only weight percentage, for comparison • Run no-enzyme controls and dosage ladders • Define inactivation strategy before scale-up
QC Checks for Performance and Release
A dairy enzyme complex should be approved against measurable technical outcomes, not only marketing descriptions. For lactose hydrolysis, common QC checks include residual lactose by HPLC or enzymatic assay, glucose/galactose increase, sweetness shift, and freezing point or solids impact where relevant. For cheese enzymes, QC may include flocculation time, curd firmness, moisture, fat in dry matter, protein recovery, whey clarity, yield, melt, stretch, shredability, bitterness, and ripening profile. For yogurt, evaluate fermentation curve, final pH, viscosity, syneresis, flavor, and shelf-life stability. Incoming material QC should verify lot number, appearance, activity specification, microbial limits, carrier declaration, storage condition, and expiration date. Production release should connect enzyme lot, dosage, batch record, processing temperature, residence time, and finished-product results to support traceability and continuous improvement.
Measure conversion, yield, texture, and sensory endpoints • Track enzyme lot and dosage in the batch record • Verify incoming activity and storage condition • Compare pilot data with plant-scale performance
Supplier Qualification and Cost-in-Use
Procurement should qualify dairy enzyme suppliers with the same discipline used for other critical processing aids. Request a current COA for each lot, a technical data sheet with activity method and recommended use conditions, an SDS for handling and storage, allergen and gluten statements, country of origin, manufacturing site information where available, shelf-life data, and change-control expectations. Cost-in-use should be calculated from delivered cost per activity unit, required dosage, processing time saved, yield improvement, waste reduction, rework reduction, and any impact on filtration, fermentation, cleaning, or heat treatment. The lowest price per kilogram may not be the best value if activity is low or inconsistent. Before commercial approval, run bench trials, pilot validation, and at least one controlled plant trial to confirm performance under real residence time and mixing conditions.
Request COA, TDS, SDS, allergen statement, and traceability • Compare cost per activity unit, not only price per kilogram • Validate at bench, pilot, and plant scale • Include supplier change-control in purchasing terms
Technical Buying Checklist
Buyer Questions
No. In this industrial context, gluten dairy enzymes refers to enzyme preparations selected for dairy manufacturing while supporting gluten-control documentation needs. It is not medical advice and does not describe consumer digestive enzyme use. Dairy processors should evaluate activity profile, carrier system, allergen statements, process conditions, and finished-product performance before approving any enzyme for cheese, yogurt, or milk processing.
A microbial coagulant within a dairy enzyme program may be evaluated as a rennet alternative when the product standard, labeling approach, and local regulations allow. It must be validated in the specific cheese make procedure. Key checks include flocculation time, curd firmness, moisture, yield, whey clarity, bitterness, melt, stretch, and ripening behavior compared with the incumbent coagulant.
At minimum, request a lot-specific COA, technical data sheet, SDS, allergen statement, gluten-control or gluten-status statement, storage conditions, shelf life, origin information, and traceability details. For supplier qualification, also ask about activity test methods, change-control procedures, packaging format, recommended handling, and support for pilot validation. Documentation should be reviewed before plant trials and again before commercial purchasing.
Start with the supplier’s activity-based recommendation, then run a structured dosage ladder against a no-enzyme control. For lactase, measure residual lactose and sensory changes. For coagulants, track flocculation, cut time, curd strength, moisture, and yield. For proteases or lipases, include bitterness, texture, and shelf-life checks. Final dosage should balance performance, consistency, process time, and cost-in-use.
The main risk is assuming that bench performance will transfer directly to production. Plant-scale mixing, temperature gradients, residence time, milk variability, culture interaction, and heat treatment can change enzyme performance. A controlled pilot or plant trial should capture actual pH, temperature, hold time, dosage, lot number, and QC results. Scale-up approval should be based on repeatable finished-product data.
Related Search Themes
digestive enzymes for dairy intolerance, best digestive enzymes for gluten and dairy intolerance, digestive enzymes for gluten and dairy, hilma dairy gluten digestive enzymes, gluten free dairy free digestive enzymes, gluten dairy digestive enzymes
Dairy Enzyme Complex for Research & Industry
Need Dairy Enzyme Complex for your lab or production process?
ISO 9001 certified · Food-grade & research-grade · Ships to 80+ countries
Frequently Asked Questions
Are gluten dairy enzymes the same as consumer digestive enzymes?
No. In this industrial context, gluten dairy enzymes refers to enzyme preparations selected for dairy manufacturing while supporting gluten-control documentation needs. It is not medical advice and does not describe consumer digestive enzyme use. Dairy processors should evaluate activity profile, carrier system, allergen statements, process conditions, and finished-product performance before approving any enzyme for cheese, yogurt, or milk processing.
Can Dairy Enzyme Complex be used as a rennet alternative?
A microbial coagulant within a dairy enzyme program may be evaluated as a rennet alternative when the product standard, labeling approach, and local regulations allow. It must be validated in the specific cheese make procedure. Key checks include flocculation time, curd firmness, moisture, yield, whey clarity, bitterness, melt, stretch, and ripening behavior compared with the incumbent coagulant.
What documents should a dairy enzyme supplier provide?
At minimum, request a lot-specific COA, technical data sheet, SDS, allergen statement, gluten-control or gluten-status statement, storage conditions, shelf life, origin information, and traceability details. For supplier qualification, also ask about activity test methods, change-control procedures, packaging format, recommended handling, and support for pilot validation. Documentation should be reviewed before plant trials and again before commercial purchasing.
How should dosage be optimized in a dairy formulation?
Start with the supplier’s activity-based recommendation, then run a structured dosage ladder against a no-enzyme control. For lactase, measure residual lactose and sensory changes. For coagulants, track flocculation, cut time, curd strength, moisture, and yield. For proteases or lipases, include bitterness, texture, and shelf-life checks. Final dosage should balance performance, consistency, process time, and cost-in-use.
What is the main risk when scaling from lab to plant?
The main risk is assuming that bench performance will transfer directly to production. Plant-scale mixing, temperature gradients, residence time, milk variability, culture interaction, and heat treatment can change enzyme performance. A controlled pilot or plant trial should capture actual pH, temperature, hold time, dosage, lot number, and QC results. Scale-up approval should be based on repeatable finished-product data.
Ready to source?
Turn This Guide Into a Supplier Brief Request a Dairy Enzyme Complex TDS, COA, SDS, and pilot trial recommendation for your cheese, yogurt, or milk processing line.
Contact Us to Contribute